Our specialized team with over 20 years oncology experience helps to ensure rapid patient recruitment, sound biomarker identification, regulatory expertise, and protocols strategically designed for smooth transition to Phases II /III and beyond to commercialization.
The foundation built with our experienced Early Phase Oncology team can help chart your course to future success. Choose Emerald Clinical for the operational expertise, forward thinking strategy, and data integrity needed to move to regulatory submission without preventable delay.
End-to-end program acceleration begins with a collaborative partnership.
Early Phase studies determine the future of your program. Emerald Clinical Trials can guide you through the critical steps, keeping your study focused, relevant, and scientifically aligned. Our therapeutic expertise, operational diligence, and geographic reach help de-risk development from PK-PD modeling to Phase III and beyond.
Gain speed and strategic leverage for a seamless transition to Phases II/III and NDA start.
Our approach ensures a quicker site start-up and higher adherence to protocol, leading to more consistent patient recruitment and retention. We prioritize quality, reliability, and agility across logistics, laboratories, and data management, while enhancing scientific input and fostering strong investigator engagement. Each study design is carefully aligned with real-world clinical practice, supported by ongoing oversight and active involvement from senior leadership to drive successful outcomes.
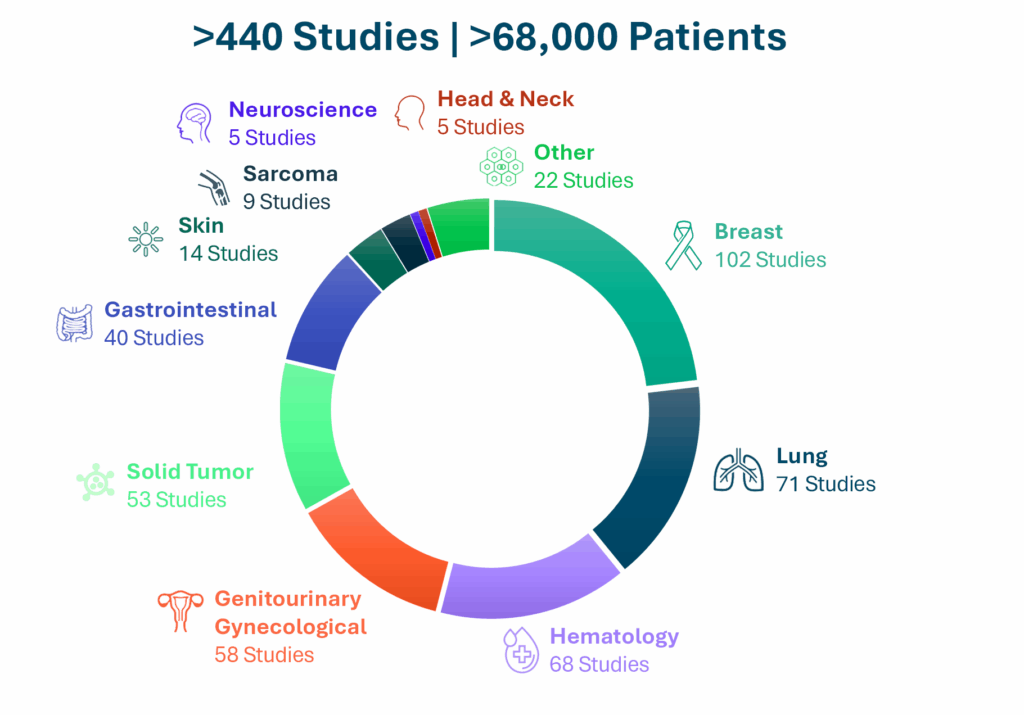
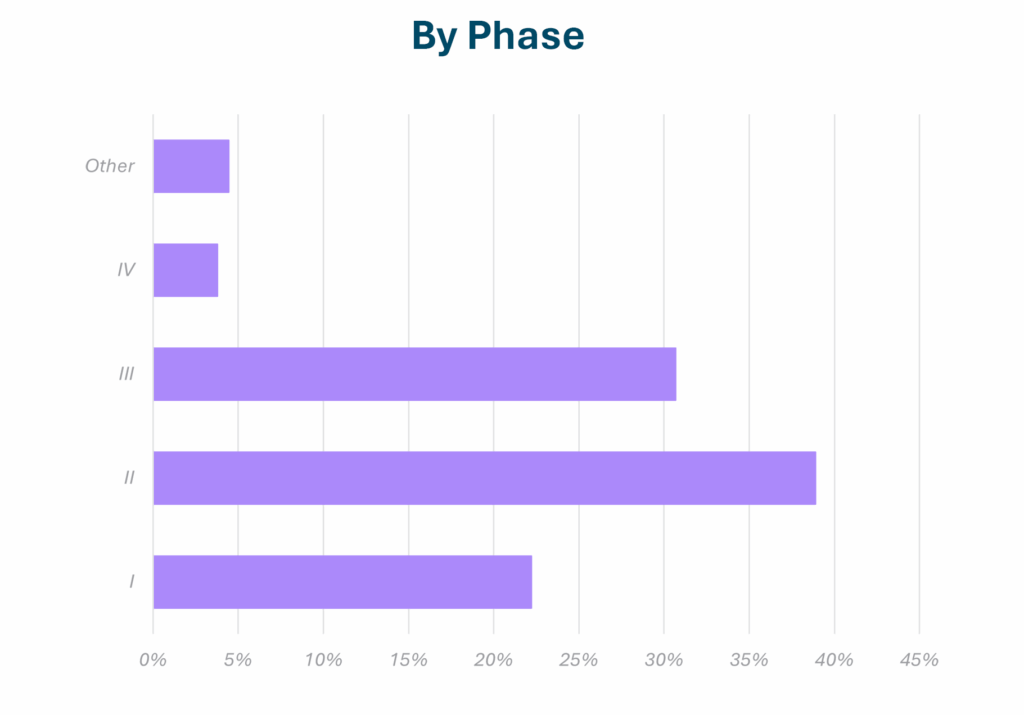
Specialized CRO Services Across Cancer Types
Our Team of Early Phase Experts Streamlines the Lifecycle of Your Program
Our experienced Oncology Phase I team extends beyond Phase I and works closely with late-phase teams to ensure a seamless transition into pivotal trials. Aligning on endpoints and positioning therapies for long-term success helps anticipate downstream challenges.
A tightly integrated, cross-functional communication network keeps studies agile, transparent, and aligned from first contact through final data lock. A single point of contact ensures all functional leads are engaged early and remain synchronized, promoting rapid issue resolution, timeline alignment, and continuous feedback loops. Collaborative tools and real-time reporting dashboards give sponsors direct visibility into progress, risks, and milestones.
The Australian Advantage
Australia offers generous R&D tax incentives up to a 43.5% rebate on eligible expenditures, and studies can begin without an IND. A strong clinical infrastructure, adherence to ICH-GCP standards, and FDA-accepted data make it an attractive destination. Emerald Clinical supports smooth transitioning to a US-based Phase II study.
Supporting you in accelerating study start-up: A conversation with Emerald Clinical Trials, Global Head, Early Phase
Emerald Clinical Trials excels in Early Phase Oncology studies by providing a collaborative, patient-centered approach. Our team offers comprehensive support throughout the study lifecycle, ensuring scientific and operational objectives align, facilitating seamless transitions to later-phase trials, and enhancing commercialization. With strong relationships with key opinion leaders (KOLs), sites, and vendors, we ensure efficient patient recruitment, robust data management, and quick trial start-ups. Our integration of adaptive trial designs and proactive risk management minimizes delays, while their focus on patient retention and regulatory compliance strengthens trial outcomes.
Read more
Why Emerald Clinical?
We help shape your Early Phase strategy to deliver clinical, regulatory, and commercial success.
Strategic Design Matters
Turn innovation into therapies with insight, experience, and trusted guidance from our dedicated, experienced Early Phase Oncology team.
Your Gateway to the Future
We start with the finish line in mind to navigate you seamlessly through Phases II/III and ultimately to NDA.
Transition Across the Globe
Our strong APAC ties, KOL relationships, and boots on the ground around the world enable seamless global expansion.
Full-Service Capability
Integrated services range from regulatory strategy, medical monitoring, biostats, scientific advisory, and site operations.
De-Risk, Accelerate, and Amplify
Prevent delays from day one. Every week you gain speed, is a week earlier to market — and earlier to market can mean millions in savings.
Real Collaboration — Long Term Value
Emerald Clinical succeeds when you succeed. Our goal is yours — get effective treatments to patients everywhere faster.
Meet the Experts Leading the Way
Tamara Murdock
Global Head of Early Phase
- 25+ years of clinical research experience in pharmacology and therapeutic areas including oncology, metabolic disorders, cardiology, and dermatology.
- A distinguished leader in Early Phase, she has worked in a Phase I unit in Australia and with Australian-based CROs.
- As Global Head of Early Phase Research, she is dedicated to ensuring that studies are conducted with precision, efficiency, and the highest scientific and ethical standards.

Rachel Condjella
Therapeutic Strategy Director, Oncology
- 18+ years of oncology experience and 6+ years in clinical project management.
- Has managed projects from inception and development to operational initiation through closeout.
- Areas of expertise include implementation and management of basket/umbrella trials, FIH trials, precision medicine for targeted treatments, and multiple types of cancer.
- Has authored publications contributing to the identification of new therapeutic and diagnostic options for cancer patients.
- Participated in the creation and presentation of data at international conferences.

For timely execution of your Phase I trial with proactive groundwork laid for Phase II site collaboration, choose Emerald Clinical’s experienced Early Phase specialists.
Find out more here about our Early Phase capabilities

